聴講希望の方は、ご所属、お名前、連絡先等記してメールにてお問い合わせください。
オープンセミナー事務局: rish-center_events@rish.kyoto-u.ac.jp
開催日当日午前10時までにご連絡ください。
要旨
Grasses are potent biomass resources for the sustainable generation of biomass-based fuels and materials due to their high growth rate and biomass productivity. However, biorefinery processes such as production of fermentable sugars from plant biomass are hindered by lignins, a major component in the plant secondary cell walls. Understanding lignin structure and biosynthesis may facilitate bioengineering and molecular bleeding approaches for reducing lignin recalcitrance and improve the biomass utilization processes. The biosynthesis and structure of grass lignins are substantially different from those of lignins in other typical vascular plants (i.e., dicots and gymnosperms). Relatively recently, it was discovered that grass lignins uniquely incorporate a flavonoid tricin, apart from monolignols, canonical lignin monomers commonly found in all vascular plants. However, not much was known about the enzymes involved in tricin biosynthesis, functions of tricin-incorporated lignin (tricin-lignin or flavonolignin), and how manipulating tricin biosynthesis impacts cell wall chemotype and biomass utilization properties in grasses. Here, I summarize our recent findings on identification of tricin biosynthetic genes and characterization of their loss-of-function rice mutants with emphasis on their cell wall chemotypes and biomass utilization properties. Rice mutant deficient in flavone synthase II (FNSII) produced lignins devoid of tricin and heterologously incorporated with a non-canonical flavonoid monomer, naringenin. A novel apigenin 3′-hydroxylase/chrysoeriol 5′-hydroxylase (A3′H/C5′H; CYP75B4) involving in both 3′ and 5′-hydroxylation steps crucial for tricin biosynthesis was identified, and its mutant lignins were likewise depleted in tricin and incorporated with non-canonical apigenin monomer. By contrast, the close homolog of A3′H/C5′H, flavonoid 3′-hydroxylase (F3′H; CYP75B3), appeared to contribute insignificantly in tricin-lignin biosynthesis but was the major 3′-hydroxylase for the parallel biosynthetic pathway leading to the formation of soluble flavone C-glycosides. On the other hand, rice mutant deficient in 5-hydroxyconiferaldehyde O-methyltransferase (CAldOMT) was depleted in both tricin-lignins and the canonical syringyl (S)-lignins, suggesting its bifunctional roles in both tricin and monolignol biosynthetic pathways. All these tricin-depleted mutants displayed improvement in biomass digestibility. As the tricin biosynthetic enzymes are highly conserved in grasses, they might serve as potent molecular targets for bioengineering to improve biomass utilization properties for biorefinery applications in other grass species.
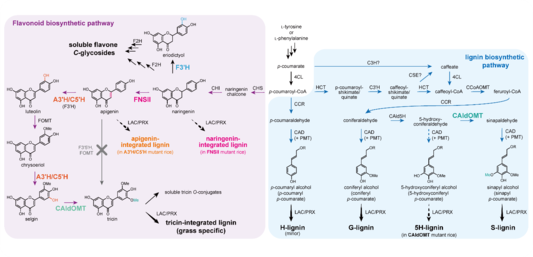 Fig. 1 Proposed tricin and monolignol biosynthetic pathways in rice. Enzymes characterized in this study are labelled with different colours.
Fig. 1 Proposed tricin and monolignol biosynthetic pathways in rice. Enzymes characterized in this study are labelled with different colours.
印刷用PDFファイル(300 729 バイト) | ページ先頭へもどる
2020年9月1日作成


